“Everything you need to know.”
(+91) 9446697551
sascanmeditech@gmail.com
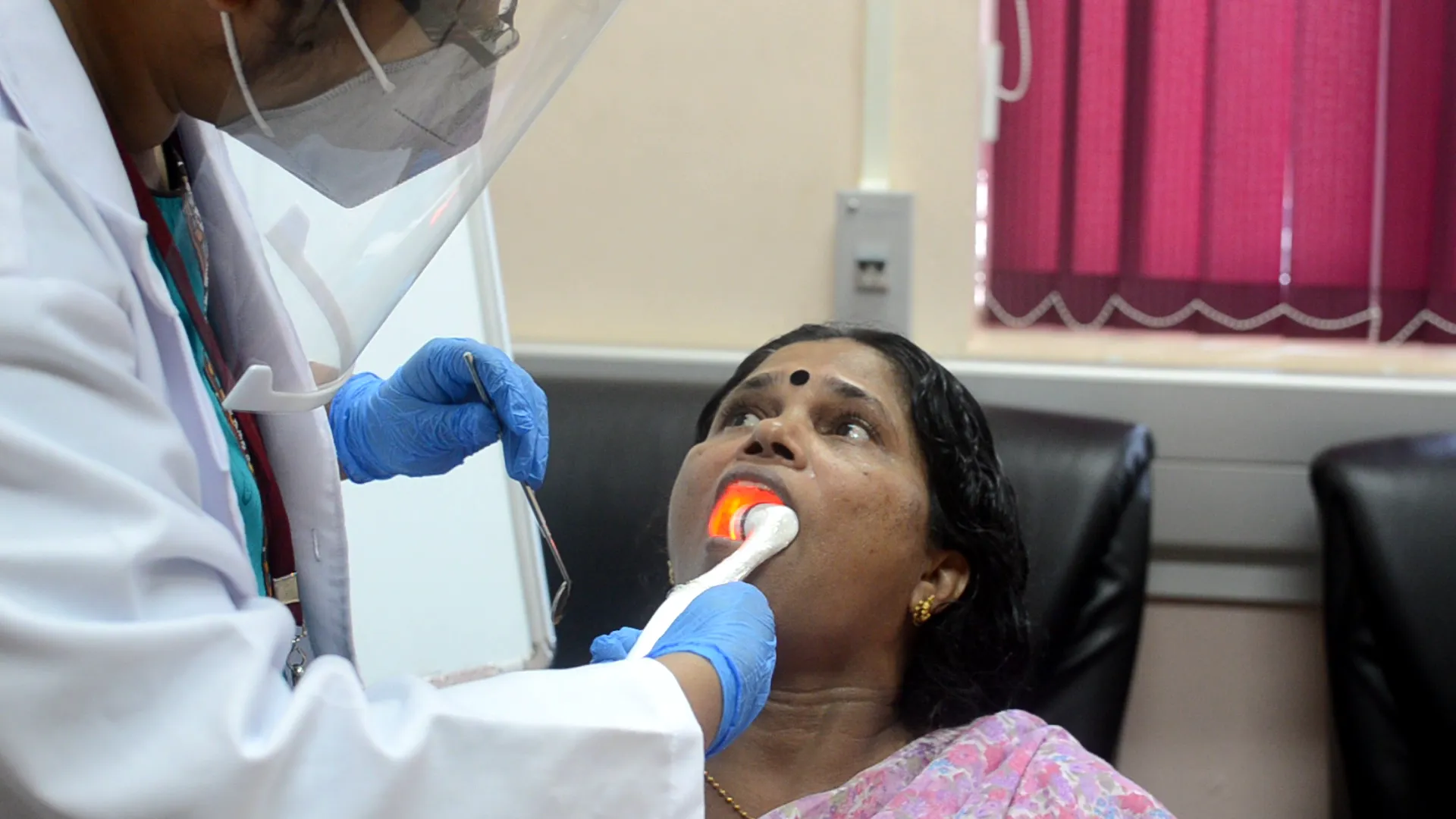
OralScan is a handheld imaging device that consists of multiple LEDs emitting violet, green, and red wavelengths to illuminate the oral cavity tissue and a monochrome camera for image capture. The device has the ability to screen and detect pre-cancers of the oral cavity and guide the clinician to the most malignant site for biopsy. The biopsy guidance feature of the device will improve the accuracy of disease diagnosis and patient care.
The patient would be asked to wash his/her mouth with water and/or chlorhexidine. After cleaning the mouth with a cotton swab to remove the presence of saliva, the site in the oral cavity will be screened for any abnormality using OralScan in video mode with violet light illumination. If any abnormal lesions are seen visually, or on the display screen in PC or monochrome modes, a detailed investigation is conducted following image capture and analysis using a cloud-based ML algorithm.
No, this is a non-invasive device that will not touch any body parts and the intensity of LED light used is well within safe limits.
Oralscan records the diffusely reflected light from the tissue which has signature information about oxygenated hemoglobin absorption changes in abnormal tissue. to increase the accuracy of the device it records the autofluorescence in blue light.
No, the device uses low-power LEDs in the visible range.
The device should be calibrated each time the room light conditions change. It is suggested to calibrate the device every day before initiating the screening.
If during screening the device gets disconnected, the probe should be removed from the port and then reconnected. On the software, a pop-up window will appear and the ‘retry’ option should be selected.

Innovating medical technology for better healthcare. Committed to quality, safety, and clinical excellence.
© 2026 Sascan Meditech Pvt. Ltd. All rights reserved.